Electron configuration periodic table qustbh
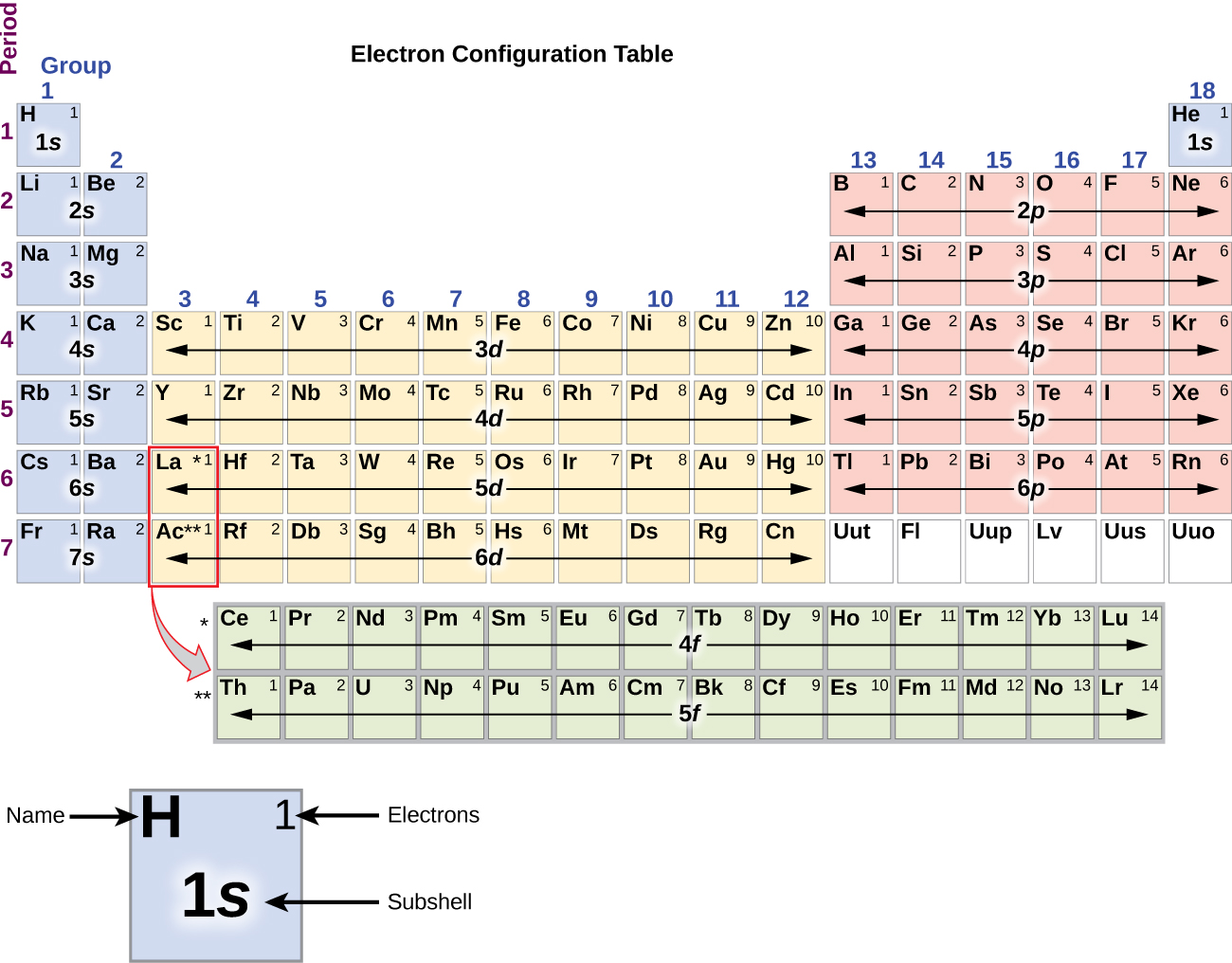
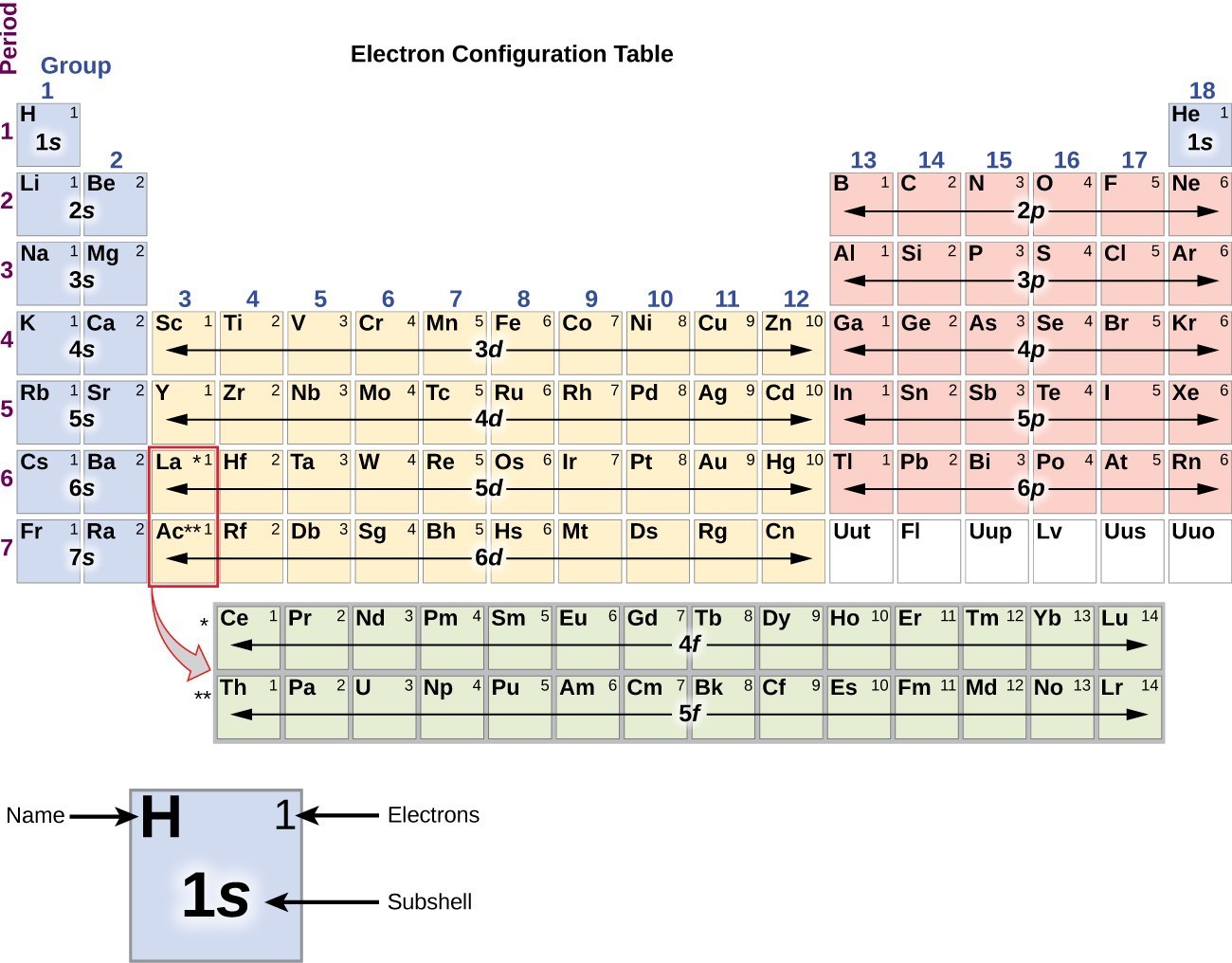
Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. The electron configuration for the first 10 elements. H #1s^1# He #1s^2# Li #1s^2 2s^1# Be #1s^2 2s^2# B #1s^2 2s^2 2p^1#

Electron configuration example for phosphorus Science, Chemistry
1). You can effortlessly find every single detail about the elements from this single Interactive Periodic table. 2). You will get the detailed information about the periodic table which will convert a newbie into pro. 3). You will also get the HD images of the Periodic table (for FREE).

Write the general outer electronic configuration of `s`,`p`,`d` and
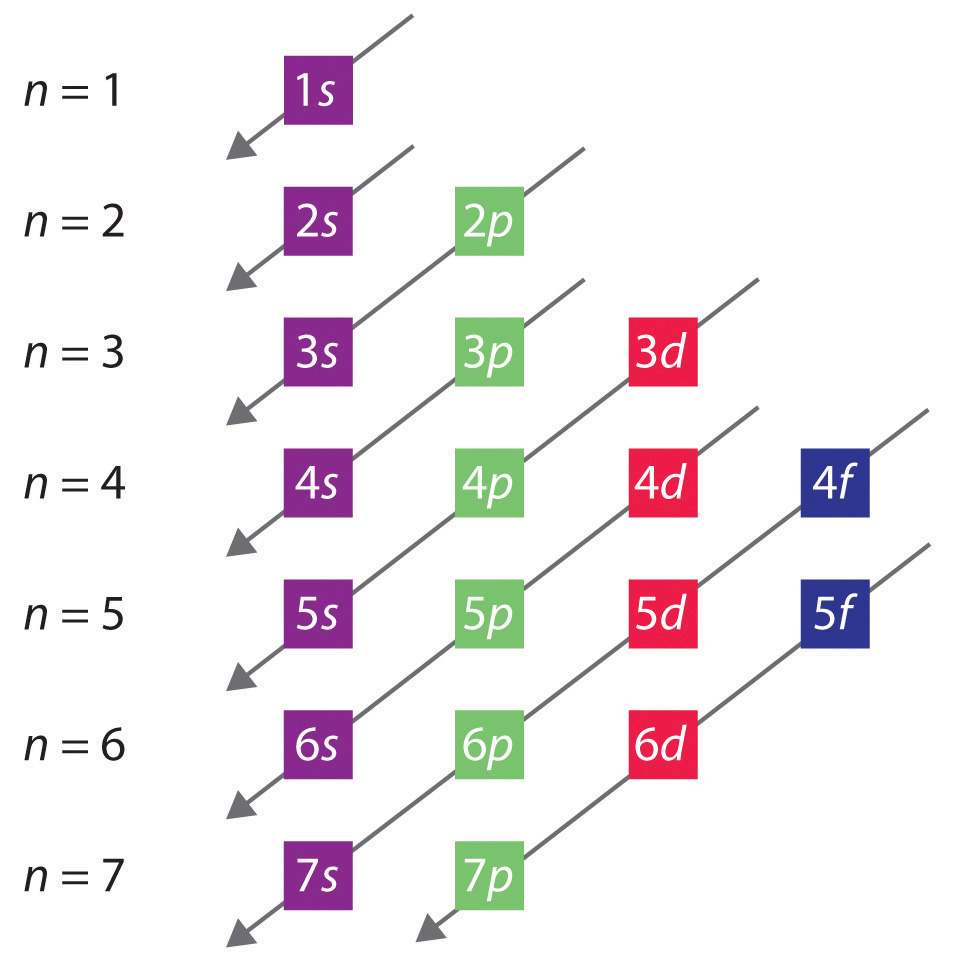
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 6.25 ): The number of the principal quantum shell, n,
Electron Structure ALevel Chemistry Revision Notes
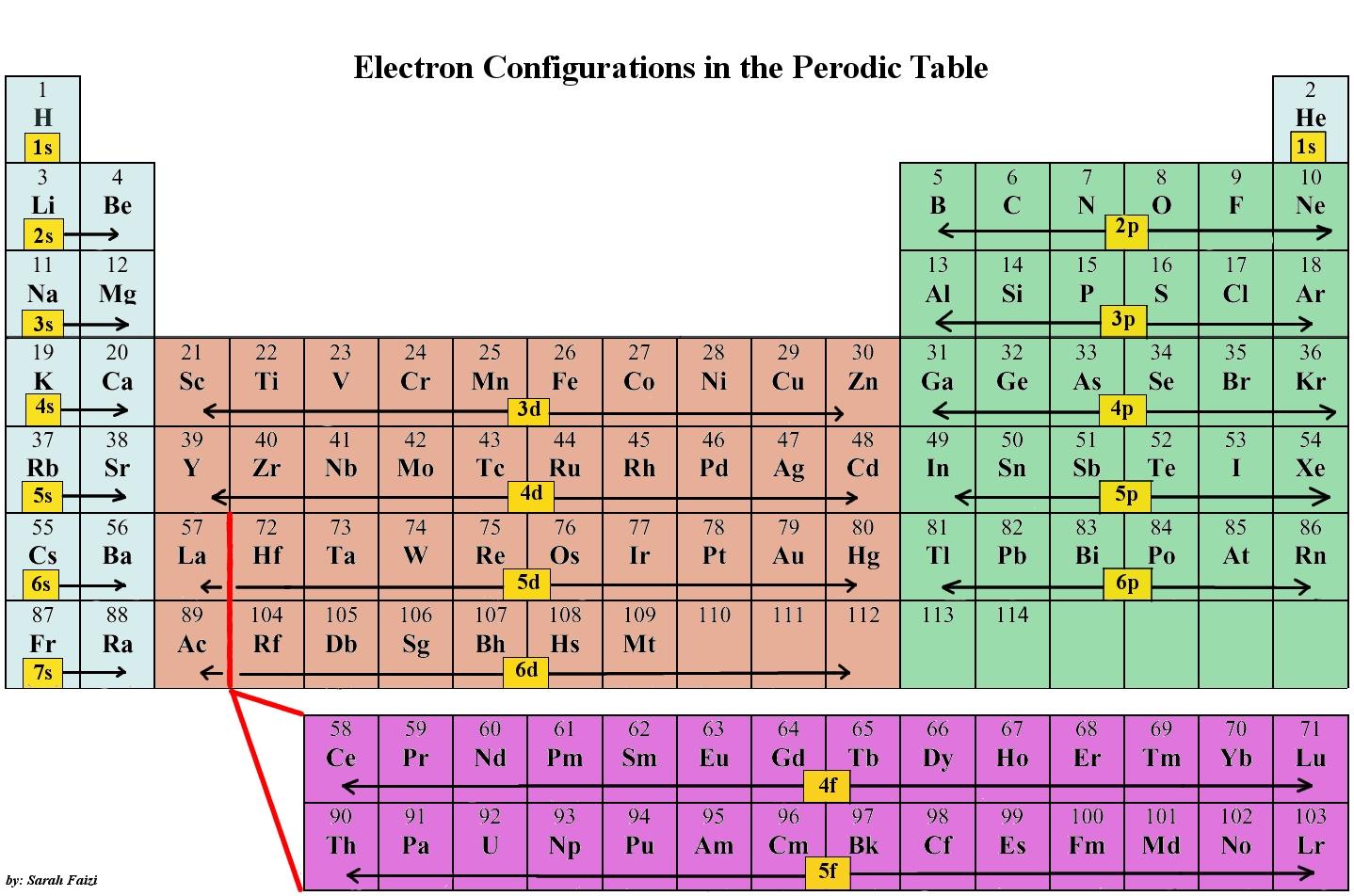
The electronic configuration of cations is assigned by removing electrons first in the outermost p orbital, followed by the s orbital and finally the d orbitals (if any more electrons need to be removed). For instance, the ground state electronic configuration of calcium (Z=20) is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2.
Electron Configuration Periodic Table Elements Chemistry
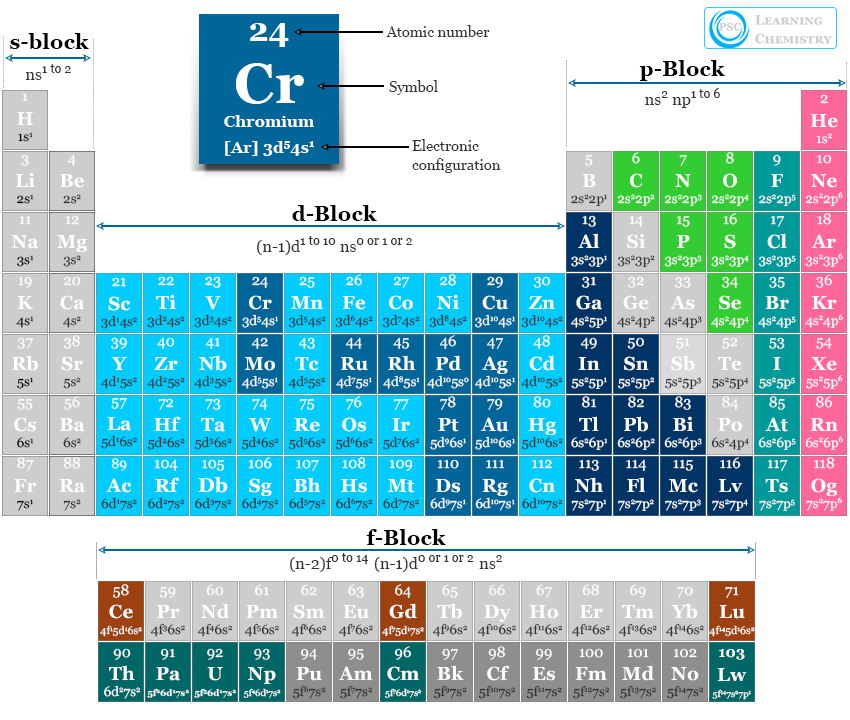
Electron Configuration Notation: -shows the arrangment of electrons around the nucleus of an atom. - helps chemist understanding how elements form chemical bonds. - can be written using the period table or an electron configuration chart. How to Write the Electron Configuration for Phosphorus (P)

Electronic Configurations Intro Chemistry LibreTexts
0:00 / 1:45 A step-by-step description of how to write the electron configuration for Phosphorus (P). In order to write the P electron configuration we first need to kn.
Structura electronica a Atomilor (Configurații de Electroni) Chimie
This page shows the electron configurations of the neutral gaseous atoms in their ground states. For each atom the subshells are given first in concise form, then with all subshells written out, followed by the number of electrons per shell. For phosphorus (element 15) as an example, the concise form is [Ne] 3s 2 3p 3.
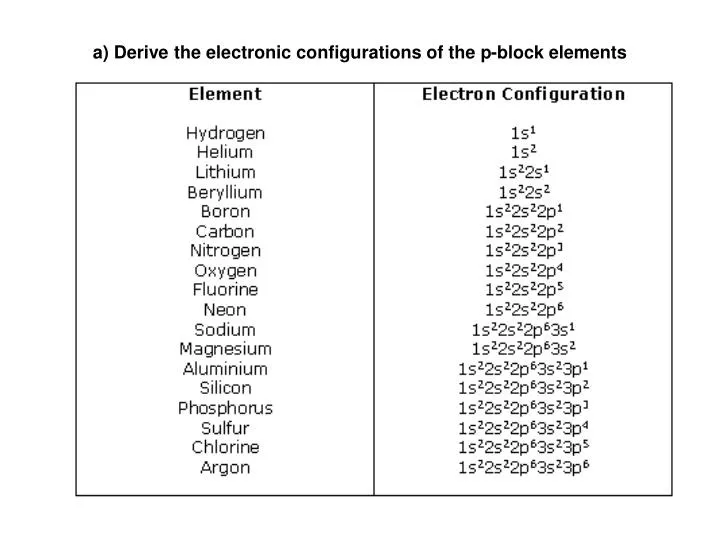
PPT a) Derive the electronic configurations of the pblock elements
In this video we will write the electron configuration for Phosphours (P) and P 3- (the Phosphide ion). We'll also look at why Phosphorous forms a 3- ion and.

Electronic Configuration Of Elements Trick (s,p,d,f) Pattern Class11
This electron configuration is written as 1 s2 2 s1. The next element is beryllium, with Z = 4 and four electrons. We fill both the 1 s and 2 s orbitals to achieve a 1 s2 2 s2 electron configuration: When we reach boron, with Z = 5 and five electrons, we must place the fifth electron in one of the 2 p orbitals.

Electronic Configuration How To Write Electron ConfigurationChemistry
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 3.1.2 3.1. 2 ): The number of the principal quantum shell, n, The letter that designates the orbital type (the subshell, l ), and
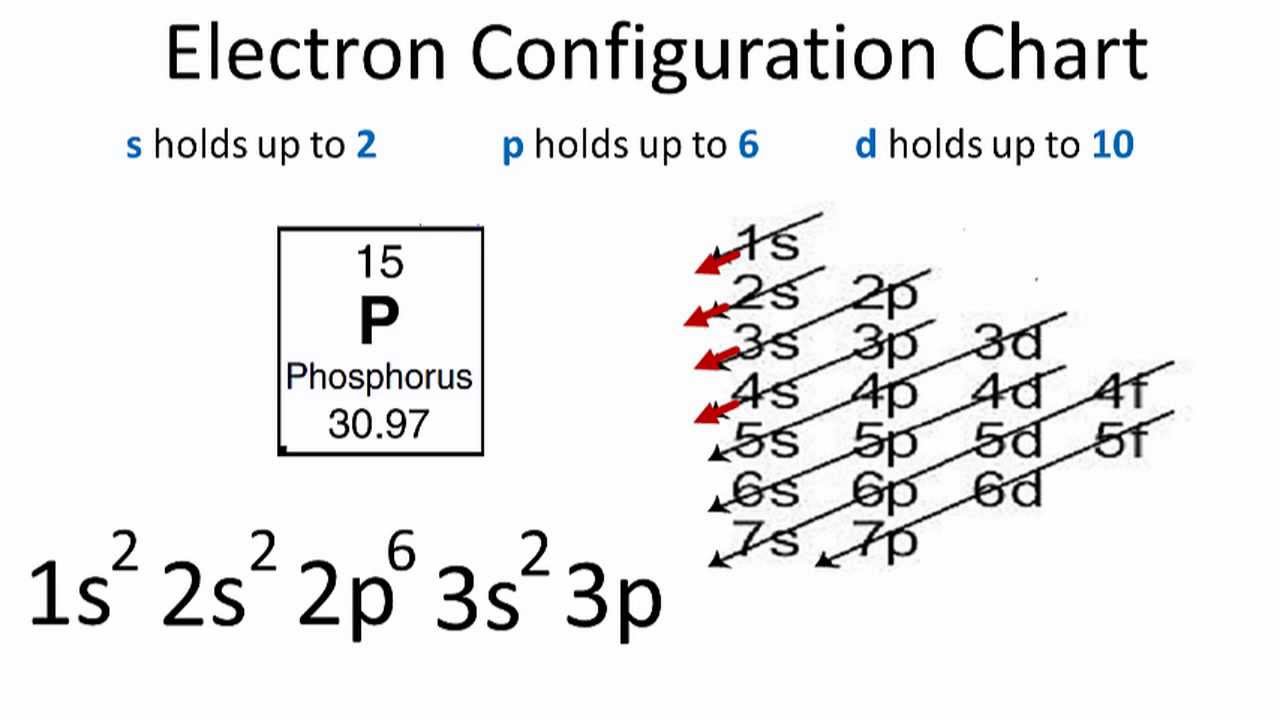
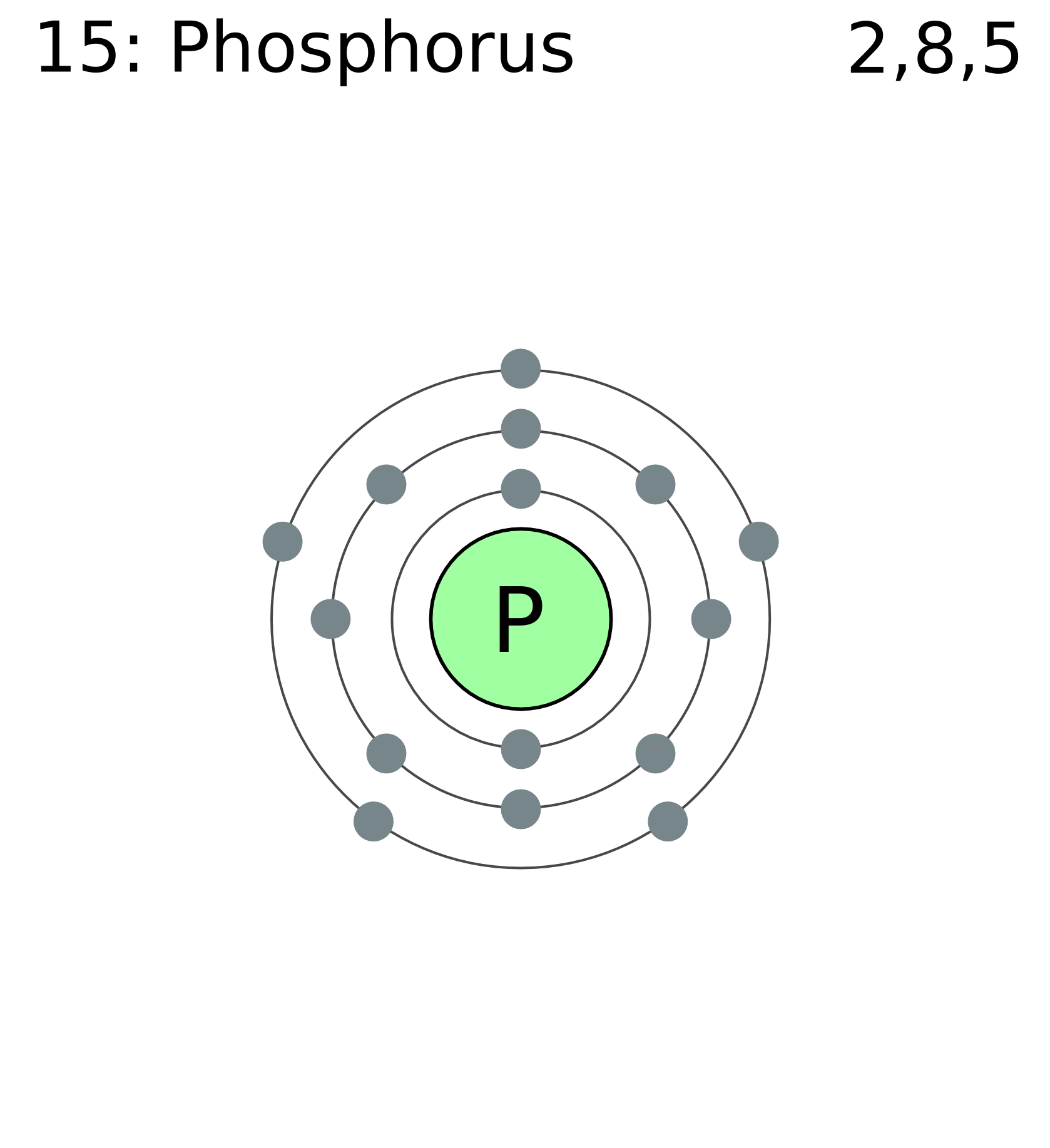
Phosphorus Electron Configuration (P) with Orbital Diagram
The valence electron configuration for phosphorus is s^2 p^3. Phosphorus has an electron configuration of 1s^2 2s^2 2p^6, 3s^2 3p^3. Phosphorus is found in group 15, the other non-metals on the periodic table. Phosphorus is in the 3rd energy level, (3rd row) and 3rd column of the 'p' block 3p^3. The valence electrons are always found in the 's' and 'p' orbitals of the highest energy level of.
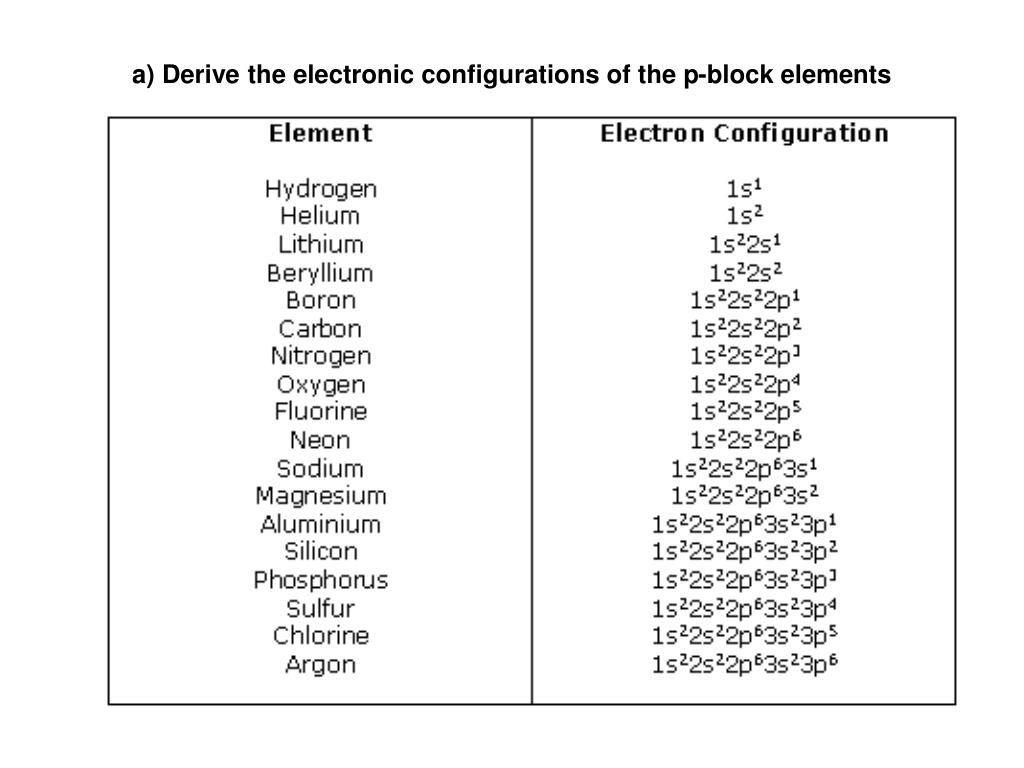
PPT a) Derive the electronic configurations of the pblock elements
In this case, 2+2+6+2+6+2+10+6+2+1= 39 and Z=39, so the answer is correct. A slightly more complicated example is the electron configuration of bismuth (symbolized Bi, with Z = 83). The periodic table gives the following electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p65s2 4d10 5p6 6s2 4f14 5d10 6p3.

Electronic Configuration of a Atom Atomic Structure Electronic
This electron configuration calculator will instantly show you the distribution of electrons in the orbitals of any periodic element you choose. Typically, you need at least 8 steps to determine the electron configuration, starting with finding the atomic number by looking at the list of orbitals and understanding the notation.
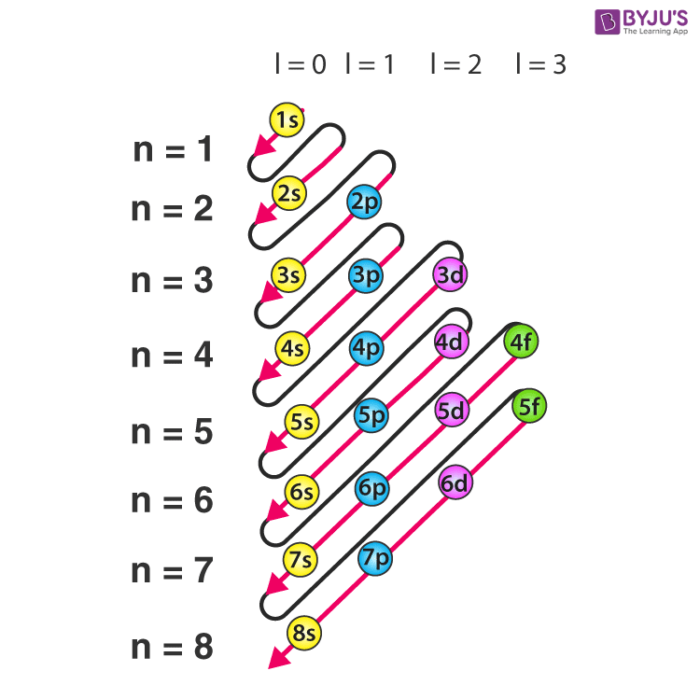
Electronic configuration detailed explanation, orbital filling
The arrangement of electrons in phosphorus in specific rules in different orbits and orbitals is called the electron configuration of phosphorus. The electron configuration of phosphorus is [ Ne] 3s 2 3p 3 , if the electron arrangement is through orbitals. Electron configuration can be done in two ways.
Question 9267e Socratic
AboutTranscript. Electron configurations describe where electrons are located around the nucleus of an atom. For example, the electron configuration of lithium, 1s²2s¹, tells us that lithium has two electrons in the 1s subshell and one electron in the 2s subshell. Created by Sal Khan.
Phosphorus Electron Configuration (P) with Orbital Diagram
The shorthand electron configuration for Phosphorus is [Ne] 3s 2 3p 3. The electron configuration for the Phosphide ion (P 3-) is 1s 2 2s 2 2p 6 3s 2 3p 6. The number of valence electrons available for the Phosphorus atom is 5. Phosphorus is situated in Group 15th or 5A and has an atomic number of 15.